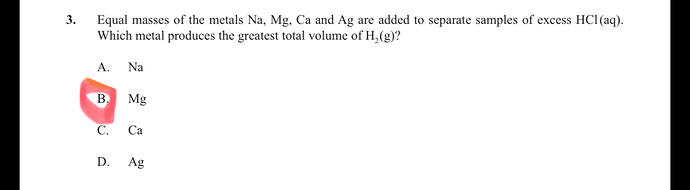Why is B the correct answer?
Answer credited to @Asafmen
Magnesium is less reactive and electropositive than Calcium, yet more reactive/electropositive than Silver and Sodium.
While not more reactive in it’s 1st ionization energy, it is more reactive with HCl in regards to the fact it uptakes 2 Cl ions and not 1 (as would be for sodium), hence it goes further with the reaction. So a 2nd period yield will yield more bonds with the chloride ions and will make more Hydrogen gas out of the dissociation of the acid.
Note that Silver and Sodium have only 1^+ ion while Magnesium and calcium have 2^+, making HCl’s dissociating and bonding to the metal to the metal at a greater rate.
HCl dissociates into Cl^- and H^+ ions which can then turn into H_2 gas.
What may have been missed here is the fact that we have similar masses, not moles. Per mass, Magnesium has more atoms than calcium. So there are more atoms binding to chloride ions and making Hydrogen gas by dissociation of HCl.
An atom of Calcium is heavier than an atom of Magnesium. (Heavier atomic mass and molar mass.)
For more: