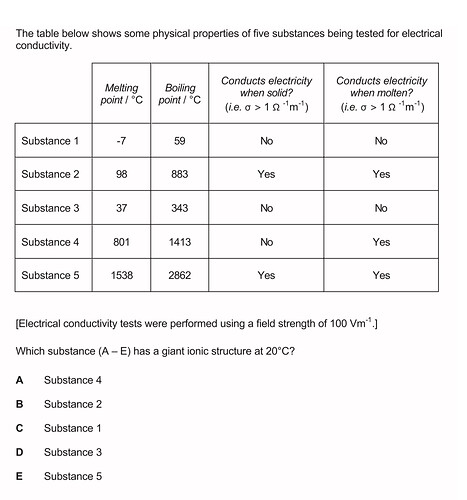
Hi, the answers to the question is A (substance 4)
I couldn’t decide between substance four or five.
Hi!
An example of a substance with a giant ionic structure is table salt (NaCl).
It doesn’t conduct electricity when solid, only when molten or dissolved in a solution.
It also has a high melting point and boiling point since it has a lot of electrostatic attractions between the positive and negative ions, we need to add a lot of energy (heat) to break
With this in mind only substance 4 will fit
2 Likes