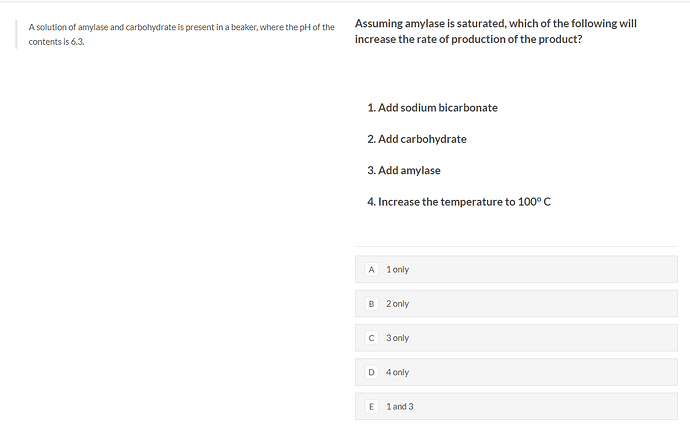
hi
is the answer E)?
- amylase functions in the small intestine, with an optimal alkaline pH. Adding bicarbonate raises the pH closer to the optimum, so the rate of the enzymes would increase
- the enzymes are saturated, so adding additional product can’t increase the rate
- adding enzymes could increase the rate
- although increased temperature can increase the rate, most enzymes in the body are denatured after 40°C, so at 100° they definitely will be denatured, reducing the rate
no, the answer is C
third one which says add amylase, how could that work?
Hi
Given that the amylase is saturated which means if there is 1 mol of amylase, all 1 mol of amylase enzyme molecules are occupied with its substrate( carbohydrates) in the solution.
So, adding none other than the enzyme itself won’t increase the rate of reaction, adjusting the ph and temperature to none other than the optimal points for the corresponding enzyme might increase the rate of reaction as well, unfortunately the optimal points for pH and temperature are not given in this problem. so we will need to choose the statement 3 only which is the option C
What rules out Option 1 though? Thought the initial explanation by Juliette made sense?
Right. but adding sodium bicarbonate might increase the rate of reaction both in theory and reality ( I agree with Juliette) but the optimal pH and temperature for amylase aren’t given in THIS QUESTION. So, u can leave that out XD
Ahh I get you in the context of this particular question we cant say that! ![]()
![]()