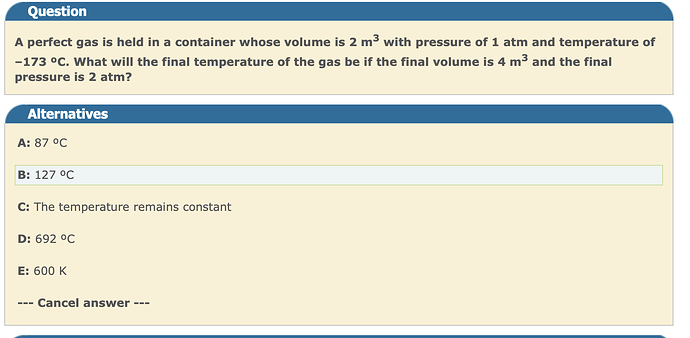
hi!
Could the answer be D°?
Here is my reasoning:
Using the ideal gas law PV=nRT (with nR constant in this question), we can see P is directly proportional to T, and V is directly proportional to T as well.
Let’s first double P:
2PV=nR2T
Let’s double V:
2P2V=nR4T
So final temperature is -173 * 4 = - 692°C
Does anyone know why i’m not finding + 692°C? Thanks!