hi!
The correct answer is C)
How can we solve this question?
Thanks!
Ringer liquid is a physiological solution used to keep isolated animal organs alive. It contains the following ions in the indicated concentrations:
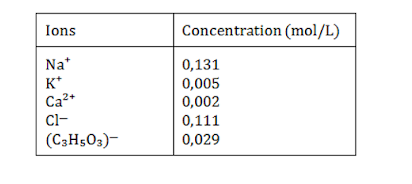
What salts must be dissolved in the water?
A) CaCl2 ; KCl ; NaC3H5O3
B) NaCl ; KCl ; Ca(C3H5O3)2
C) NaCl ; CaCl2 ; KCl ; NaC3H5O3
D ) NaCl ; CaCl2 ; KCl ; KC3H5O3